Over 4,000 providers across the U.S. are empowered to easily and affordably target dermatological conditions for their patients.
Enhance adherence by offering combined ingredients in one medication.
Boost overall efficiency and profitability by eliminating the administrative burden of managing insurance claims, allowing staff to focus more on patient care.
Spare patients from waiting in pharmacy lines and avoid medication substitutions to ensure they receive the medication they deserve at a more accessible price.
Choose medications without potentially harmful inactive ingredients and allergens commonly found in comparable branded and generic drugs*.
SKNV’s hassle-free prescribing experience increases patient loyalty and drives referrals. Additionally, we help you target core skin conditions. Depending on the needs of your practice and applicable state regulations, SKNV medications are available through two main models:
You can purchase inventory, immediately provide the medication to your patient at the time of their visit, and monetize your practice, all while offering a great experience to your patient.
You can prescribe from your EMR to send our customized medications directly to your patient’s home with no sales tax (in most states) and no shipping cost to your patients.
Branded & Generic Medicines
may contain inactive ingredients that are potential allergens or irritants for certain patients, as determined by the provider*
SKNV
medications without potentially harmful inactive ingredients and allergens, for certain patients and as determined by prescribers
SKNV
pharmacy for sensitive skin nationwide
SKNV
patients prescribed SKNV Rx every month for their sensitive skin
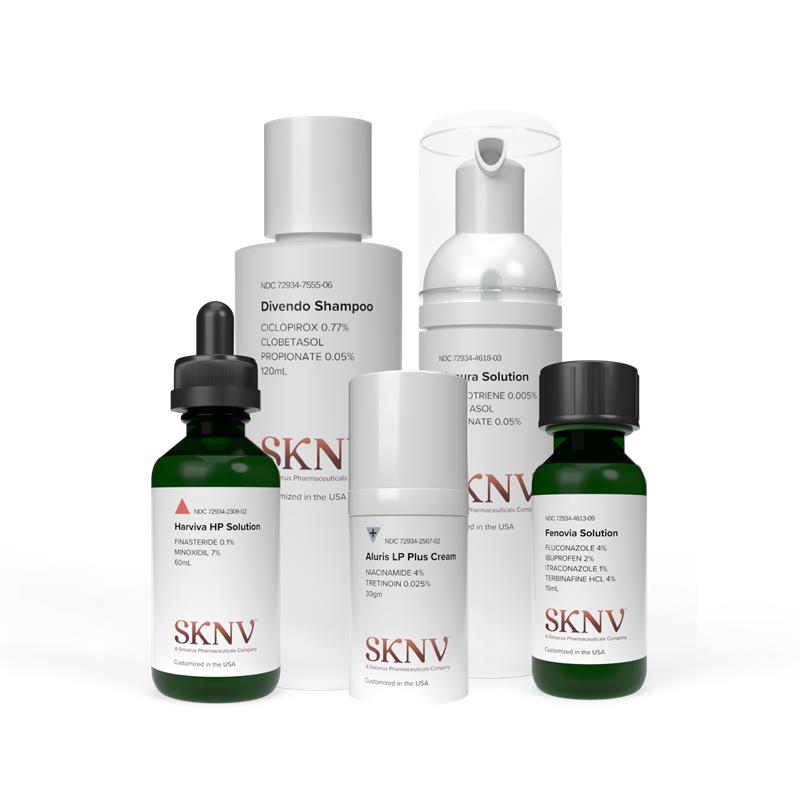
SKNV was born from the realization that the U.S. insurance industry was failing dermatology patients.
SKNV provides reliable medications at a great price point. Following stringent quality assurance measures, our formulations are tailored to your skin needs and without potential harmful excipients found in comparable branded and generic drugs, for certain patients as determined by the provider.*
Spencer Malkin, SKNV CEO and Co-Founder