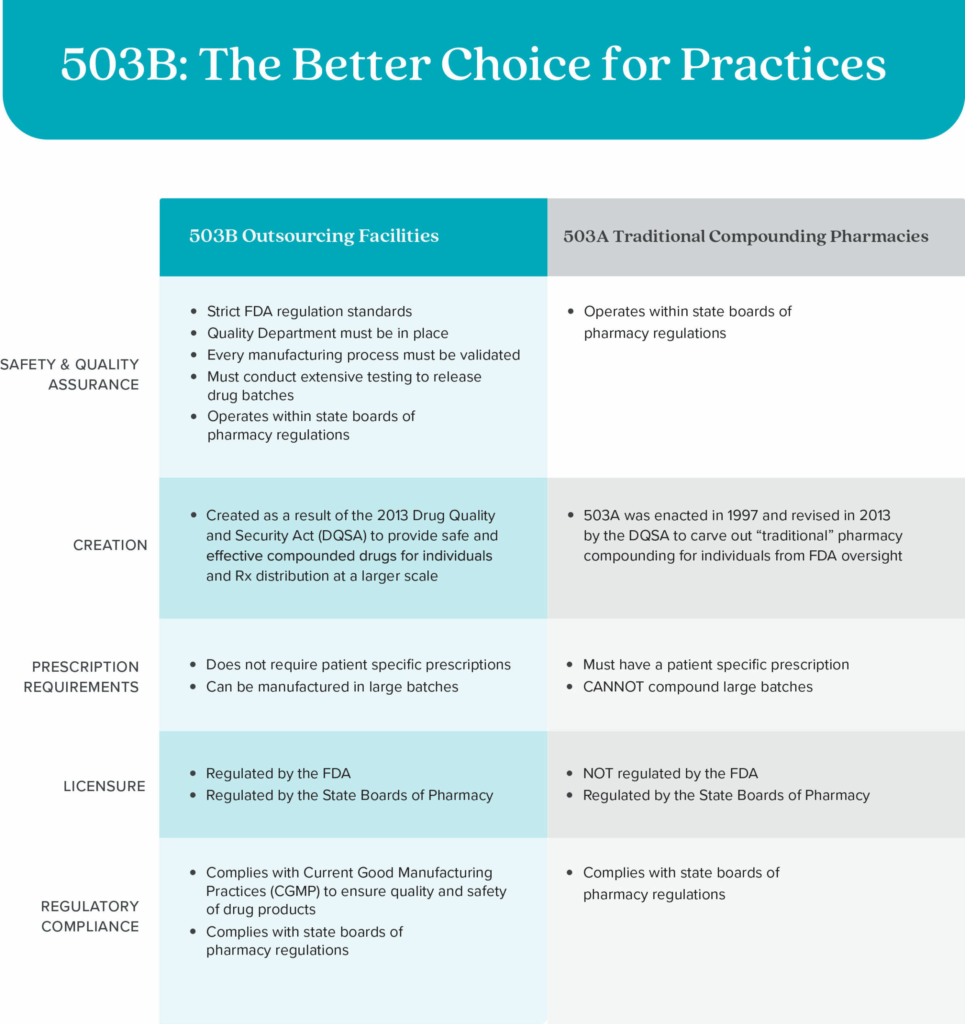
503B Outsourcing Facilities are not the same as 503A Compounding Pharmacies. 503A Compounding Pharmacies are not registered or regularly inspected by the FDA, cannot compound in large batches, and do not allow practices to dispense from their office.
In 1938, Congress passed a set of laws giving authority to the U.S. Food and Drug Administration (FDA) to oversee the safety of food, drugs, and cosmetics. Dubbed the Federal Food, Drug, and Cosmetic Act (FD&C Act), this gave the legal framework within which the FDA operates.
In 2013, a 503A company, New England Compounding Center, caused a tragic and deadly meningitis outbreak due to their contaminated medications. This major incident highlighted significant lapses in the quality control and safety standards of compounded drugs.
After this incident, Congress passed the Drug Quality and Security Act (DQSA), which amended the FD&C Act to grant the FDA more authority to regulate and monitor the manufacturing of compounded drugs. This legislation aimed to prevent future tragedies by ensuring higher standards of safety and quality in the compounding pharmacy industry. A new category of FDA-regulated companies was born, and this is why SKNV was founded.